-
- au
Ici vous trouverez le papier de Karim Chouchane
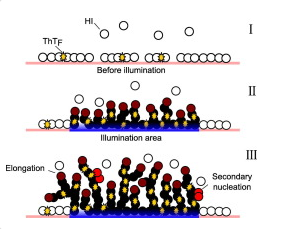
"Insulin is known to form amyloid aggregates when agitated in a hydrophobic container. Amyloid aggregation is routinely measured by the fluorescence of the conformational dye thioflavin T, which, when incorporated into amyloid fibers, fluoresces at 480?nm. The kinetics of amyloid aggregation in general is characterized by an initial lag-phase, during which aggregative nuclei form on the hydrophobic surface. These nuclei then lead to the formation of fibrils presenting a rapid growth during the elongation phase. Here we describe a novel mechanism of insulin amyloid aggregation which is surprisingly devoid of a lag-time for nucleation. The excitation of thioflavin T by visible light at 440?nm induces the aggregation of thioflavin T-positive insulin fibrils on hydrophobic surfaces in the presence of strong agitation and at physiological pH. This process is material surface-induced and depends on the fact that surface-adsorbed insulin can bind thioflavin T. Light-induced insulin aggregation kinetics is thioflavin T-mediated and is based on an energy transfer from visible light to the protein via thioflavin T. It relies on a constant supply of thioflavin T and insulin from the solution to the aggregate. The growth rate increases with the irradiance and with the concentration of thioflavin T. The supply of insulin seems to be the limiting factor of aggregate growth. This light-induced aggregation process allows the formation of local surface-bound aggregation patterns."


