-
- au
Ici vous trouverez le papier de C. Verrier
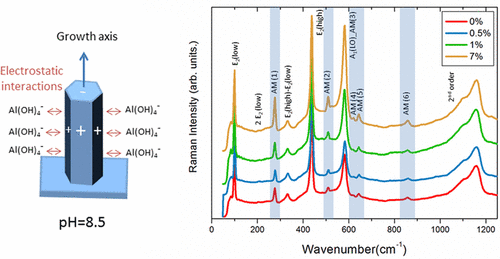
"Mastering the properties of ZnO nanowires grown by the low temperature chemical bath deposition (CBD) is of crucial importance but is still challenging. We show that the shape, dimensions, and doping of ZnO nanowires can simultaneously be tuned by the addition of aluminum nitrate in the standard chemical system using zinc nitrate, hexamethylenetetramine, and ammonia in aqueous solution. The formation and doping mechanisms of ZnO nanowires are thoroughly investigated by combining chemical, structural, and optical analyses with in situ pH measurements correlated with thermodynamic simulations. We reveal that the electrostatic interactions of Al(OH)4– complexes with the positive m-plane sidewalls of ZnO nanowires at a given pH favor their adsorption as capping agents, reducing the radial growth and promoting the elongation, while favoring the aluminum uniform incorporation. Importantly, the aluminum doping is found to be thermally activated above the low temperature of 200 °C under oxygen atmosphere, as indicated by the occurrence of six related additional modes in the range of 200–900 cm–1 in temperature-dependent Raman spectroscopy. These findings show that CBD using aluminum nitrate is of high potential for tuning both the morphology of ZnO nanowires and their physical properties via the aluminum doping, which paves the way for their more efficient use into sensing, electronic, and optoelectronic devices on both flexible and rigid substrates."
DOI: 10.1021/acs.jpcc.6b11104


